Enzymes (2H2O2 --> 2H2O + O2)
1. Background Information
a. Enzymes
~ Catalysts increase the rate of reaction without being consumed by the reactions
~ Enzymes lower the activation energy of the reaction
~ Enzymes are SPECIFIC to their substrate because the shape of the active site conforms to the shape of the substrate
~ Denaturation of the enzyme can occur from pH or temperature changes
~ Co-factors = non-protein attachments to the enzyme's active site that help maintain it's shape (ex. vitamins)
~ Competitive Inhibition mimics the substrate and blocks the active site, whereas non-competitive inhibition binds to another section on the enzyme, causing the shape of the active sight to change ~ Allosteric Regulation either causes activation by stabilizing the enzyme shape, or can cause inhibition by destabilizing the enzyme shape
~ Cooperativity are enzymes that can have multiple active sites, so induced fit at one active sight may cause stabilization of other active sites on the enzyme
b. Catalase is a biological enzyme that breaks down hydrogen peroxide and turns it into less harmful substances such as oxygen and water. Cells produce catalase; it's used to detoxify the cell. If the cell doesn't produce enough catalase, the cell will die.
c. Enzyme-Substrate interaction: 2H2O2 ---> 2H2O + O2
a. Enzymes
~ Catalysts increase the rate of reaction without being consumed by the reactions
~ Enzymes lower the activation energy of the reaction
~ Enzymes are SPECIFIC to their substrate because the shape of the active site conforms to the shape of the substrate
~ Denaturation of the enzyme can occur from pH or temperature changes
~ Co-factors = non-protein attachments to the enzyme's active site that help maintain it's shape (ex. vitamins)
~ Competitive Inhibition mimics the substrate and blocks the active site, whereas non-competitive inhibition binds to another section on the enzyme, causing the shape of the active sight to change ~ Allosteric Regulation either causes activation by stabilizing the enzyme shape, or can cause inhibition by destabilizing the enzyme shape
~ Cooperativity are enzymes that can have multiple active sites, so induced fit at one active sight may cause stabilization of other active sites on the enzyme
b. Catalase is a biological enzyme that breaks down hydrogen peroxide and turns it into less harmful substances such as oxygen and water. Cells produce catalase; it's used to detoxify the cell. If the cell doesn't produce enough catalase, the cell will die.
c. Enzyme-Substrate interaction: 2H2O2 ---> 2H2O + O2
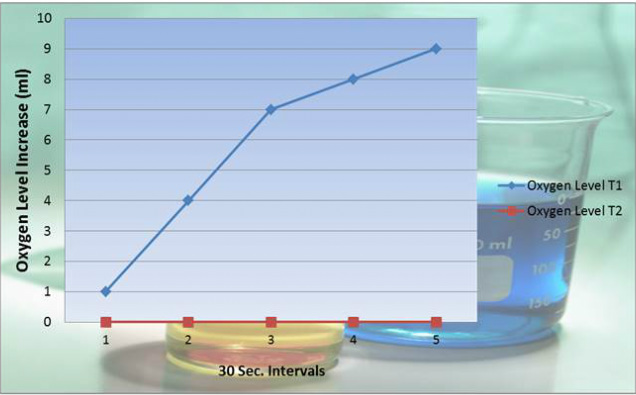
Oxygen Level Part A (T1):
Water Level: Full
Temperature: 17.6 degrees Celsius
Oxygen Part 2(T2):
Water Level: Full
Temperature: 17.6 degrees Celsius
Variable: 2 Drops of HCL
When the Hydrochloric Acid was added to the experiment in Part 2 (T2), the enzyme reaction was stopped. All of the enzymes were denatured and no longer functioned correctly to break down the hydrogen peroxide in the apparatus.
The intial rate of the reaction started at 0.033. Then gradually rose to a 0.1, and ends at a steady rate of 0.033.

The apparatus was filled with: 10 ml of hydrogen peroxide and 4ml of catalase (turnip slurry). Then the mixture was quickly sealed and held underneath the water. The reaction took place in the apparatus, and the oxygen traveled through the reaction chamber and into the graduated cylinder. Oxygen would travel up the graduated cylinder and "push" the water down the tube and into the water bath (clear pan).

(From left to right) 2 droppers, Graduated Cylinder, Ring Stand with Clamp, Water Bath (Clear Pan), 10ml Graduated Cylinder, and the Reaction Chamber


